Celularity Reports First Quarter 2022 Financial Results and Provides Corporate Update
Ongoing Phase 1 study of unmodified placental-derived natural killer (NK) cell therapy candidate, CYNK-001, in acute myeloid leukemia (AML) continues enrolling new cohorts to reflect adjustments to treatment regimen and dosing as outlined in December 2021
Ongoing Phase 1 study of CYNK-001 in glioblastoma multiforme (GBM)
U.S. Food and Drug Administration (FDA) granted Fast Track designation to genetically modified placental-derived NK cell therapy candidate, CYNK-101, in first line metastatic HER2/neu positive gastric and gastroesophageal junction cancers; Phase 1/2a study is ongoing
FDA granted an Orphan Drug Designation to CYNK-101 for the treatment of gastric/gastroesophageal junction cancers
FLORHAM PARK, N.J., May 16, 2022 — Celularity Inc. (Nasdaq: CELU) (“Celularity” or the “Company”), a clinical-stage biotechnology company developing placental-derived off-the-shelf allogeneic cell therapies, today announced financial results for the first quarter ended March 31, 2022, and provided a corporate update.
“We have continued to achieve multiple transformational milestones and make significant progress this year with three ongoing Phase 1 clinical trials of two investigational drugs, CYNK-001 and CYNK-101, both of which have been granted Fast Track and Orphan Drug Designations for certain indications,” said Robert J. Hariri, M.D., Ph.D., Founder, Chairperson and Chief Executive Officer of Celularity. We expect to have data readouts for all three ongoing Phase 1 programs later this year. As such, we believe we are well-positioned to continue executing our business strategy and developing cellular therapies for cancer, infectious and degenerative diseases that leverage our proprietary placental-based technology platform.”
First Quarter Clinical and Regulatory Updates
CYNK-001 for the Treatment of AML and GBM:
- CYNK-001 is Celularity’s unmodified cryopreserved human placental hematopoietic stem cell-derived NK cell therapy candidate that is enriched with CD56+/CD3- NK cells and expanded from human placental CD34+ cells. CYNK-001 is currently being investigated in two Phase 1 clinical trials, in AML and in GBM, with data readouts expected in the second half of 2022.
- Celularity continues to enroll new cohorts in both arms of the Phase 1 AML study with clinical trial protocol adjustments communicated in December 2021, which include an addition of interleukin-2 (IL-2) to the treatment regimen; a fourth dose on day 21; and an increase in the dose of NK cells.
CYNK-101 for the Treatment of Gastric Cancer:
- CYNK-101 is a novel allogeneic off-the-shelf human placental CD34+-derived NK cell product candidate that is genetically modified to express high-affinity and cleavage-resistant CD16 (FCGRIIIA) variant to drive antibody-dependent cell-mediated cytotoxicity. CYNK-101 is currently being investigated in the Phase 1 portion of a Phase 1/2a clinical trial in advanced HER2+ gastric cancer.
- In January 2022, the FDA granted Fast Track designation to CYNK-101, which is being developed in combination with standard chemotherapy, trastuzumab and pembrolizumab in first-line locally advanced unresectable or metastatic HER2/neu positive gastric/gastroesophageal (G/GEJ) adenocarcinoma.
- In February 2022, the FDA granted Orphan Drug Designation to CYNK-101 for the treatment of G/GEJ cancer.
CYCART-19 for the Treatment of B-Cell Malignancies:
- CYCART-19 is an allogeneic Chimeric Antigen Receptor (CAR) engineered human placental-derived T cell that is a potential drug candidate in B-cell malignancies.
- Preclinical data demonstrating the feasibility and functionality of expressing a CAR directed to CD19 on placental CD34+derived, cryopreserved, off-the-shelf, allogeneic CYNK cells were presented at the American Society of Hematology (ASH) Annual Meeting in December 2021.
- Celularity submitted an Investigational New Drug (IND) application to FDA to investigate CYCART-19 for the treatment of B-cell malignancies in the first quarter of 2022 and in April 2022, received email notification from FDA that it will be requesting additional information before Celularity can proceed with the planned Phase 1/2 clinical trial. Celularity anticipates a formal written communication by late May 2022 and plans to work with the FDA in an effort to resolve its questions as promptly as possible and, if the IND is cleared, commence a Phase 1/2 clinical trial of CYCART-19 in B-cell malignancies in the second half of 2022.
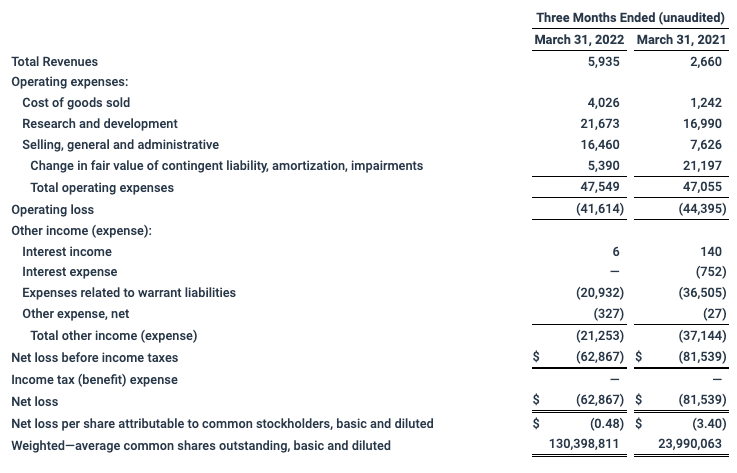
First Quarter 2022 Financial Results
- Cash and Cash Equivalents: Cash, cash equivalents and marketable securities were $48.0 million as of March 31, 2022, compared to $37.2 million as of December 31, 2021. In March 2022, we amended and restated certain warrants to reduce the exercise price per share to $3.50 per share, among other items, following which the holders exercised such warrants in full for cash for approximately $46.5 million and we issued the holders an aggregate 13,281,386 shares of our Class A common stock.
- Total Revenues: Total revenues were $5.9 million for the first quarter of 2022, compared to $2.7 million for the first quarter of 2021. This increase in revenues was primarily driven by sales activity from supply and distribution agreements for Celularity’s degenerative disease products.
- Research & Development (R&D) Expenses: R&D expenses were $21.7 million for the first quarter of 2022, compared to $17.0 million for the first quarter of 2021. The increase was primarily driven by higher clinical development expenses.
- Selling, General & Administrative (SG&A) Expenses: SG&A expenses were $16.5 million for the first quarter of 2022, compared to $7.6 million for the first quarter of 2021. The increase in SG&A expenses was primarily caused by higher expenses related to being a public company, such as stock-based compensation, insurance and consulting fees.
- Net loss: Net loss for the first quarter of 2022 was $62.9 million, or $(0.48) per share for both basic and diluted shares. Net loss for the first quarter of 2021 was $81.5 million, or $(3.40) per share for both basic and diluted shares.
About Celularity
Celularity Inc. (Nasdaq: CELU) headquartered in Florham Park, N.J., is a clinical stage biotechnology company leading the next evolution in cellular medicine by developing allogeneic cryopreserved off-the-shelf placental-derived cell therapies, including therapeutic programs using unmodified natural killer (NK) cells, genetically modified NK cells, T-cells engineered with a CAR (CAR-T cells), and mesenchymal-like adherent stromal cells (ASCs). These therapeutic programs target indications in cancer, infectious and degenerative diseases. In addition, Celularity develops and manufactures innovative biomaterials also derived from the postpartum placenta. Celularity believes that by harnessing the placenta’s unique biology and ready availability, it can develop therapeutic solutions that address significant unmet global needs for effective, accessible, and affordable therapies.
To learn more, visit celularity.com.
Forward-Looking Statements
This press release includes “forward-looking statements” within the meaning of The Private Securities Litigation Reform Act of 1995, as well as within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. All statements other than statements of historical facts are “forward-looking statements,” including those relating to future events. In some cases, you can identify forward-looking statements by terminology such as “anticipate,” “believe,” “can,” “contemplate,” “continue,” “could,” “estimate,” “expect,” “forecast,” “intends,” “may,” “might,” “outlook,” “plan,” “possible,” “potential,” “predict,” “project,” “seek,” “should,” “strive,” “target,” “will,” “would” and the negative of terms like these or other comparable terminology, and other words or terms of similar meaning. The forward-looking statements in this press release include statements regarding anticipated data readouts from its ongoing Phase 1 clinical trials, interactions with FDA regarding the CYCART-19 IND and timing of the planned Phase 1/2 clinical trial, and statements regarding Celularity’s ability to develop therapeutic solutions that address significant unmet global needs for effective, accessible, and affordable therapies, among others. Many factors could cause actual results to differ materially from those described in these forward-looking statements, including but not limited to: the inherent risks in development of cellular therapeutics, including with respect to the development of novel cellular therapies, and the clinical trial and regulatory approval process; and risks associated with developments relating to Celularity’s competitors and industry, along with those risk factors set forth under the caption “Risk Factors” in Celularity’s annual report on Form 10-K filed with the Securities and Exchange Commission (SEC) on March 31, 2022 and other filings with the SEC. These risks and uncertainties may be amplified by the COVID- 19 pandemic and the Russian invasion of Ukraine. If any of these risks materialize or underlying assumptions prove incorrect, actual results could differ materially from the results implied by these forward-looking statements. There may be additional risks that Celularity does not presently know, or that Celularity currently believes are immaterial, that could also cause actual results to differ from those contained in the forward-looking statements. In addition, these forward-looking statements reflect Celularity’s current expectations, plans, or forecasts of future events and views as of the date of this communication. Subsequent events and developments could cause assessments to change. Accordingly, forward-looking statements should not be relied upon as representing Celularity’s views as of any subsequent date, and Celularity undertakes no obligation to update forward-looking statements to reflect events or circumstances after the date hereof, whether as a result of new information, future events or otherwise, except as may be required under applicable securities laws.

Celularity Investor Contacts:
Carlos Ramirez SVP, Investor Relations
Celularity Inc.
carlos.ramirez@celularity.com

