Celularity Presents Preclinical Data on Allogeneic Genetically-Modified Natural Killer Cells at the Society for Immunotherapy of Cancer 36th Annual Meeting
FLORHAM PARK, N.J., Nov. 15, 2021 (GLOBE NEWSWIRE) — Celularity Inc. (Nasdaq: CELU) (“Celularity”) presented preclinical data on the development of its novel allogeneic genetically modified human placental CD34+ derived NK cell therapeutic program CYNK-101. Celularity reported its findings in a poster presentation at the Society for Immunotherapy of Cancer (SITC) Annual Meeting held in Washington, D.C., November 10-14, 2021.
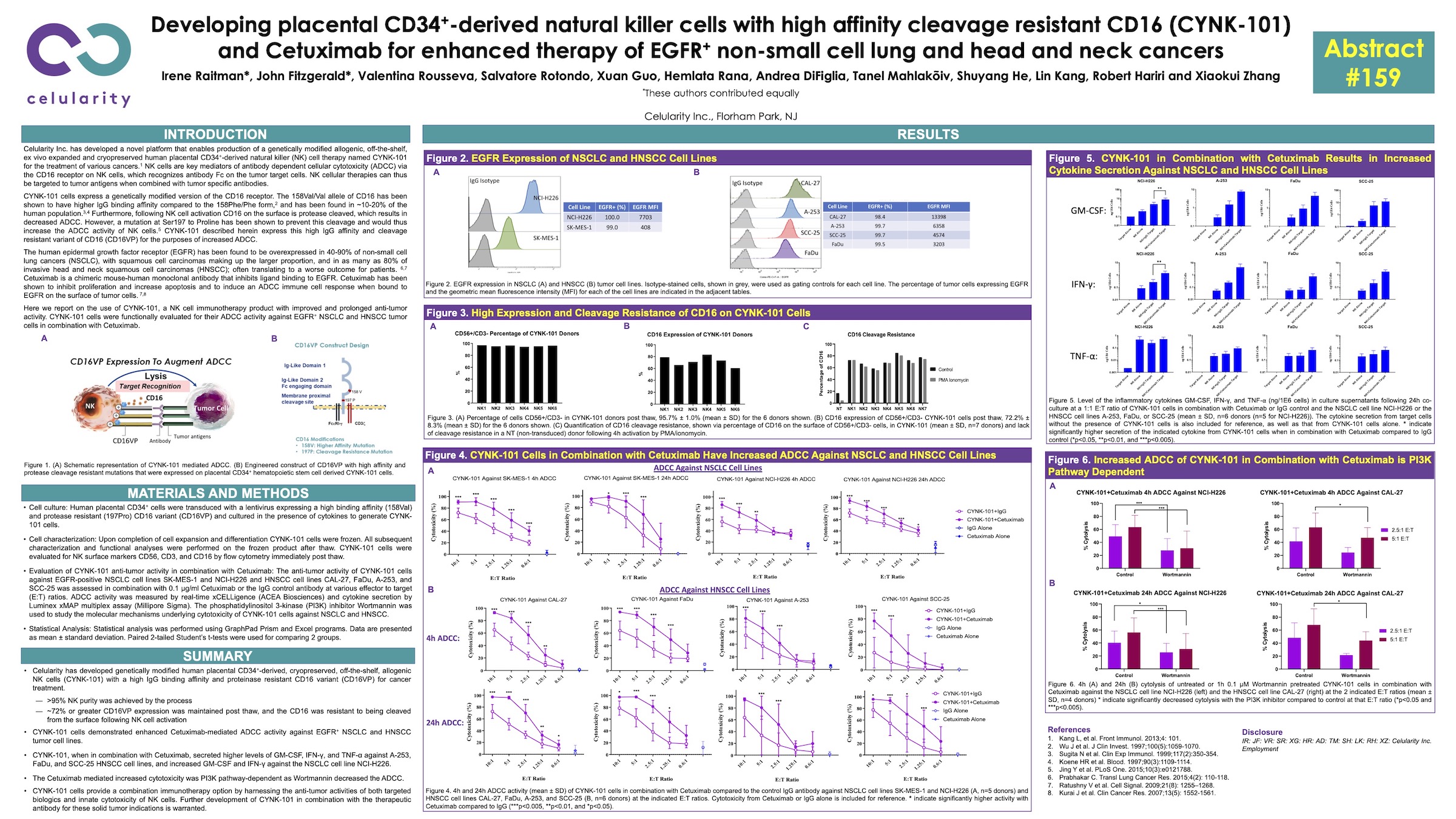
Poster 159: Developing placental CD34+-derived natural killer cells with high affinity cleavage resistant CD16 (CYNK-101) and Cetuximab for enhanced therapy of EGFR+ non-small cell lung and head and neck cancers”
Highlights: CYNK-101 is a cryopreserved off the shelf allogeneic NK cell product, overexpressing a high IgG binding affinity, proteinase cleavage resistant, CD16 variant. The results demonstrate the synergistic effect of combining CYNK-101 with Cetuximab to drive antibody dependent cellular cytotoxicity activity against EGFR+ tumors. Further development of the combinational therapy against EGFR+ non-small cell lung cancer and head and neck squamous cell carcinomas is warranted.
About Celularity
Celularity Inc. (Nasdaq: CELU) headquartered in Florham Park, N.J., is a clinical stage biotechnology company leading the next evolution in cellular medicine by developing allogeneic cryopreserved off-the-shelf placental-derived cell therapies, including therapeutic programs using unmodified natural killer (NK) cells, genetically modified NK cells, T-cells engineered with a CAR (CAR T-cells), and mesenchymal-like adherent stromal cells (ASCs). These therapeutic programs target indications in cancer, infectious and degenerative diseases. In addition, Celularity develops and manufactures innovative biomaterials also derived from the postpartum placenta. Celularity believes that by harnessing the placenta’s unique biology and ready availability, it can develop therapeutic solutions that address significant unmet global needs for effective, accessible, and affordable therapies.
To learn more, visit celularity.com.
Celularity Investor Contacts:
Carlos Ramirez SVP, Investor Relations
Celularity Inc.
carlos.ramirez@celularity.com
Alexandra Roy
Solebury Trout
aroy@troutgroup.com
Celularity Media Contact:
Jason Braco, Ph.D.
LifeSci Communications
jbraco@lifescicomms.com
- Previous PostCelularity Reports Third Quarter 2021 Financial Results and Corporate Update
- Next PostCelularity Announces FDA Clearance of Investigational New Drug Application (IND) for Natural Killer Cell Therapy CYNK-101 in First-line Advanced Her2/neu Positive Gastric and Gastroesophageal Junction Cancer